Welcome to the Bennett Lab
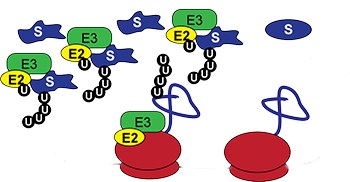
In the Bennett lab, our research is focused on understanding how the proteome is dynamically remodeled in response to cellular stress. In particular, we study mechanisms employed by the cell’s protein degradation system to buffer the cell against acute and chronic protein homeostasis stress. The ubiquitin-proteasome system is the primary cellular machinery tasked with overseeing the selective degradation of nearly every protein within the cell. One of the key functions of the ubiquitin proteasome system is to oversee the selective removal of damaged or erroneously synthesized proteins.
We utilize and develop quantitative proteomic methodologies to interrogate protein degradation function and fidelity. With these tools we can begin to quantify alterations in protein homeostasis function during the development of age-associated disorders.
The lab investigates how this quality control function of the ubiquitin proteasome system can be harnessed and exploited to protect cells from environmental and genetic insults. To accomplish this, the lab uses integrated molecular, cellular, and systems level approaches.